A quick canter through the anomalies of drug prescribing in the UK; what’s wrong with the system; and why, if you tried to design a system to make life difficult for trans kids you would be pushed to come up with a more toxic one.
Shocking, innit? All those poor vulnerable young people being turned into guinea pigs — not literally, of course! — just to satisfy the greed of a big farmer. Sorry: Big Pharma! with a capital B and a P and harm in the middle!
Down with untested drugs!
That’s why the time has come to stop feeding untested and off-label drugs to trans kids…What? No!
I’m talking about pregnant women. Worried about infection? Well, tough: no more cefalexin for you! Ditto magnesium sulphate — even if you do reckon you’re at risk from pre-eclampsia. As for high blood pressure: for you, ma’am, the nifedipine is over. You’ll just have to practice relaxation and controlled breathing instead.
At this point, some of you are likely going: has jane taken leave of her senses? Does she hate women? Why would she insist on people suffering when we have medical treatments that can reduce that suffering?
So, i confess. It’s a joke, albeit in poor taste. Or if you want the philosophy of it, it’s a reductio ad absurdum, taking the arguments of the anti-trans around the supposed ills of “off-label medication” as prescribed to trans kids and running them out into the wider medical population to see what they might mean in practice.
Consider the following, both table and graph:
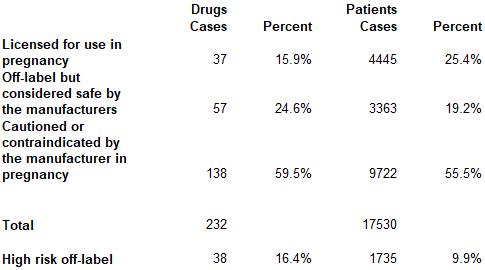
Shock! Horror! Liverpool!
The figures are from a peer-reviewed paper, Off-label prescribing during pregnancy in the UK: an analysis of 18 000 prescriptions in Liverpool Women’s Hospital, first published in the International Journal of Pharmacy Practice, in 2010. It’s not the most up to date. But there is no reason to believe it atypical of current practice. At least, not atypical of current practice in respect of two highly vulnerable cohorts: children and pregnant people.
If you think otherwise, hie thee over to Google Scholar and tap in “off-label prescribing UK”. There you will find an abundance of articles on the topic.
This is not system bug, but design feature, to which we will return in a moment.
As for the big take-out : some 75% of drugs prescribed to folks during pregnancy at one of the UK’s leading maternity facilities were “off-label”. Of those, 55% were “cautioned or contraindicated for use in pregnancy”.
Worse still, a “significant number of these prescriptions” were not just off-label, but high risk. That’s 38 drugs (16% of total) and 1735 (10%) of the total prescriptions.
As the paper somewhat elliptically comments: “prescribers need to be aware of the risks associated with these drugs and the possible legal consequences of prescribing and administering them.”
Off-label: more common than you think
It is difficult to put a definitive number to off-label prescribing. Search the literature, though, and you will regularly find estimates in the range 40% to 60%. Here’s a much more recent paper in Clinical Pharmacology and Therapeutics confirming up to 60% of child prescribing — albeit in the EU — is off-label.
Here’s another one from the Neonatal and Paediatric Pharmacists Conference 2021 confirming that “the use of unlicensed and off-label medicines is, in many circumstances, the only appropriate alternative”. Though it comes with built-in warning of risk to individual patients. Once more, the watchword is: tread careful!
Why, though, aren’t all drugs tested to the nth degree? Like, if we are going to feed some powerful chemical substance to a child, is there not some obligation on us to make sure in advance that is a) will do no harm and b) will do some good?
Let’s start with the basics. Off-label medicines are medicines being used in a way that differs from that described in the licence. Unlicensed medicines are licensed in other countries, but not the UK. These must therefore be imported.
Also, a subtle point: it is not the medicine that is off label, but its use. There are assumptions and guidelines governing how medicines are used, and these rules are broadly similar the world over. Some governing body — the FDA in the US: the HPRA in the UK — looks at the evidence around specific medicines and proposed uses, and goes public on what they think that medicine can usefully be used for.
This, though, is never a comprehensive list of uses. Every regulatory authority in the world makes it clear that GPs and a range of medical professionals are entitled to prescribe off-label/unlicensed if they feel it serves their patients best interests.
Here’s the UK guidance. Note that GP’s are exhorted to take extra care when prescribing off-label. But it is absolutely accepted they can do it and accepted that everyone does.
Improper drugs
But why are some drugs off-label/”improperly” licensed? That’s not hard to work out. Here’s how drug development works.
First, invent your drug. This may take years and cost you tens of millions. Congratulations! Now, file your patent and prepare to rake in the spondoolies.
OK. Not quite. That patent gives you 20 years clear in which to exploit your discovery. Except, in that 20 years you need to test it, prove it works and eliminate dangerous side-effects. All the time, circling shark-like, are competitors just waiting to launch a generic version of your drug. This may be identical (homologue) or very similar (analogue: think about all the fuss over whether a drug a bit like MDMA was enough similar to make it illegal a few years back)
Sometimes, competitors go live with analogues during the product life of the original, resulting in massive litigation. If you want a well-remunerated career, be a patent lawyer!
Testing, testing…
The testing phase might take you five, six, eight years. So now you have just 12 years in which to recoup your investment. The more you test, the less time you have to recoup your investment/make a profit. So, you hone it down. You test for very specific effects and don’t worry about nice-to-haves
Also, not everything can be tested ethically. That is espesh true of testing for effects (and side-effects) on children. So, you hope, you pray, as a pharma product manager that after about 5 years you have proven your drug does what it’s meant to…and doesn’t harm people
That’s it. There is no time, no scope in the system for someone to put their hand up in year 9 and go: oh….maybe this drug would be useful for x….let’s spend another few million and get it approved and licensed with just 5 years left on the patent!
So, it is left to independent researchers to turn up additional uses and, if those prove righteous, to disseminate them through medical literature and hope eventually that awareness will percolate out to GP’s and other prescribers in the system.
The issue is that the system for licensing new and useful treatments really only works well (or at all) for new drugs. A doctor can prescribe the licensed version with confidence for the indication named in the product licence, and maybe for one or two additional indications if the drug company manage to get the clinical trials done, and a label extension approved, while they still have sufficient exclusivity in the drug to recoup the cost.
Once the drug has become generic, no drug company can be incentivised to do the clinical studies to support additional indications because they are super expensive and offer precisely no commercial reward.
This is a big hole in the system, since doctors who wish to meet an urgent patient need have to prescribe ‘off label’ or on an unlicensed basis. This is unsatisfactory scientifically, exposes them to liability risk which may be uninsurable, and may technically be a breach of medicines regulations. Although nobody ever seems to object to the latter unless they have a particular axe to grind.
That is the real issue here, because mirror image to current gender critical “concerns” about off-label puberty blockers being prescribed to kids, is the Avastin case, in which media and politicians came together to demand…the prescribing of an off-label drug to some of the most vulnerable patients.
The Avastin case *
Avastin was an established bowel cancer treatment for IV use. Then, researchers discovered it could be used in diluted form for intra-vitreal use against wet age-related macular degeneration (WMD). That’s a bowel cancer drug unexpectedly found to be useful in treating a common sight condition, among older patients.
NHS doctors started using it. As there was no existing treatment and the alternative was to allow patients to go blind, the NHS carried the risk. The drug company which owned Avastin sued on the basis that diluting the drug was a breach of the medicines regulations. The reason for their interest could not possibly have been that they had a newer, ‘better’, and likely more expensive drug for WMD with which Avastin would now compete.
The media and public reaction were interesting. This was mostly along the lines of “how very dare evil big pharma let our golden oldies go blind?”. Not, as you might imagine, given the concerns over trans children: “how very dare the woke NHS give an experimental drug to patients when there’s no clinical trial data to support it?”
One-time Labour Minister, and later Baroness, Tessa Jowell suggested the drug company should do the clinical trials, with predictable response. Various people suggested the NHS should do it, but the NHS isn’t really set up for that and never has been.
Eventually, in 2019/20, the matter percolated its way up to the Court of Appeal (CoA), at which point a fudge of sorts was handed down. That is, the CoA took advantage of a loophole in the medicines regulations to say it was okay to carry on as long as the treatment was prescribed to patients on an individual bespoke basis and the dilution carried out in a hospital pharmacy. More on that judgment here.
So, everyone is now happy? Well, everyone except the drug company. However, the central problem remains: how do we know this treatment is safe? The National Institute for Health and Care Excellence (NICE) has approved the treatment on the basis of data from elsewhere (e.g., the US NIH) and recommended it over the newer drug for cost reasons.
Gender crit cakism…
Was NICE over-influenced by cost? Would we have been better off with proper clinical trial data endorsed by the regulators? It’s probably all okay. Imagine, though, what would have happened if there was a lobby group out there who had made it their mission to make sure that patients didn’t get Avastin treatment (or maybe not until after they’d gone blind) because they are ‘so worried about the lack of safety evidence’?
You could argue it either way. Though i suspect it would never be a popular headline: “The Sun Says: Let Them Go Blind!”
Nah. I don’t see that playing well, even in the cheapest of cheap seats.
Because there is a risk assessment and a trade-off, and people get that. Up to a point.
They get it when it’s gran going blind and there is a readily-available, but untested treatment available on the NHS.
They get it when it is someone giving birth, and there is an untested treatment or two that can alleviate pain, reduce risk of heart attack and Lord Knows What besides.
In both instances, i suspect, they’d want the patient alerted to risk. But that’s that. Let’s treat patients and their families as grown-ups and accept that not everything in life is certain or comes with cast-iron guarantee.
That’s a nice/NICE rational compromise and broadly people buy into it.
They get it…until we get to trans kids, and puberty blockers, which have been in use for decades and for which the straightforward clinical side-effects are well known.
So, now, there is a lobby group out there that comes at the issue with an entire arsenal of specious argument.
It’s not been tested! That, as we have seen above, is the dimmest of dim arguments, and applies to a great deal more than the humble puberty blocker.
What about the side-effects? And, true. Medicines come with side-effects. All medicines. Perhaps some pedant will drop me a line to explain how there exists some magical substance (water?) with no side-effect whatsoever. Otherwise, read the leaflet that accompanies every drug you will ever be prescribed. What do you see listed? Every single possible side-effect ever noted and that could possibly befall you.
That’s not to say they will. But they might.
Honestly, if we went only by the side-effects, we’d never prescribe a thing.
Last up, there has been an uptick lately in “concern” over the psychological consequences of taking puberty blockers. If you don’t get to experience a ‘proper’ puberty, how can you be sure you don’t want to stay cis?
In a way, there is no answer to that. Because how can you know? But it is a dangerous argument for everyone concerned. After all — and this is the long game — once you accept this argument, and the unpicking of Gillick confidence in respect of trans kids, the next step is to challenge abortion. Contraception itself.
How can you know that you don’t want this child unless you have given birth to it?
…and hypocrisy
In the end this is no more than the rankest of rank hypocrisy from folks who hate trans people and who are determined to punish trans kids by sentencing them to a lifetime of social misfittery and surgery. Because when you block puberty blockers, that is precisely what you do.
If they cared about the principles involved, cared about the dangers of off-label prescribing, they would be campaigning against the prescribing of known dangerous drugs up there in the Liverpool Women’s Hospital.
They’d be on the picket lines advocating for eye disease.
The fact that the only prescribing they appear remotely interested in is that to trans kids speaks volumes.
There is no consistency here. No principle. Just hate. And hypocrisy.
That is all.
* I am indebted, in respect of this part of today’s write-up, to a good friend who also happens to work (or at least, to have worked for many years) as an expert legal adviser to one of the UK’s evil pharma giants.

This piece was originally published on Medium.

